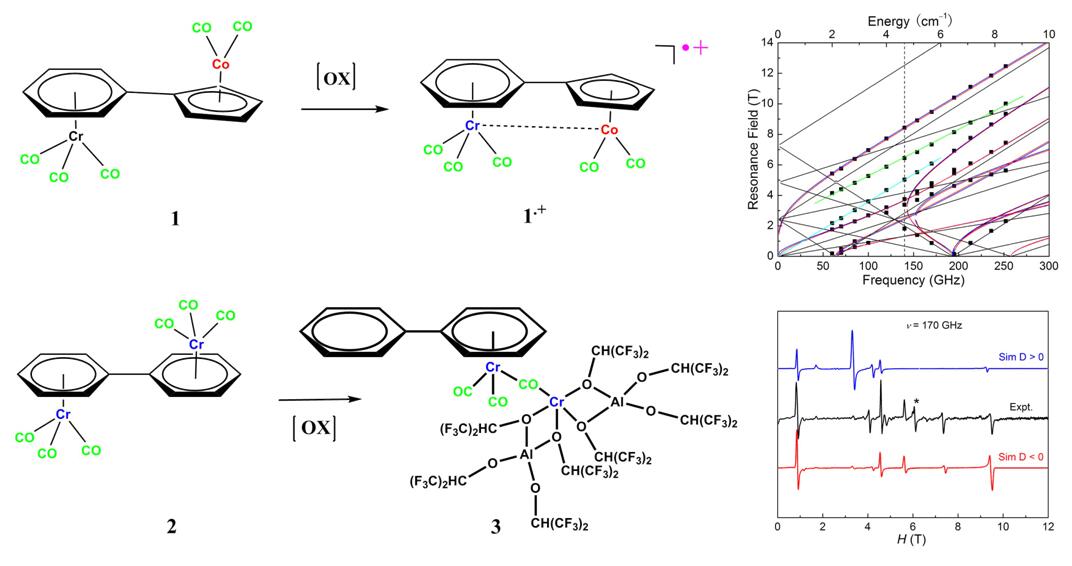
Synthesis of the carbonyl complexes and the high frequency EPR characterizations
(J. Am. Chem. Soc., 2017,DOI:10.1021/jacs.7b06795)
Background
Half-sandwich metal carbonyl complexes are a well-known class of organometallic species and are of fundamental importance in organic synthesis and organometallic chemistry. Their oxidation affords carbonyl-containing 17-electron organometallic radical cations, which are important in stoichiometric and catalytic transformations because of their high reactivity in comparison to 18-electron precursors.The bimetallic complexes are also of importance for the understanding of the nature of metal–metal bonds, however, a hetero dinuclear metalloradical containing a metal–metal hemi-bond has not been reported.
What we discover?
We report oxidation of dinuclear half-sandwich carbonyl complexes, (Cr(CO)3(η6,η5–C6H5C5H4)Co(CO)2andCr(CO)3(η6,η6–C6H5C6H5)Cr(CO)3), which led to the first example of a hetero dinuclear metalloradical which features a Cr-Co half bond and a rare example of a Cr(II) complex displaying single-molecule magnetic properties, respectively.
Why is this important?
By far, most single ion magnets (SIMs) are centered at f-block metal elements. Compared to f-block complexes, the 3d transition-metal ions can be expected to suppress the orbital contributions to the magnetism that is required to develop magnetic anisotropy. This work thus has demonstrated that oxidation of metal carbonyl complexes may lead to new species with promising physical properties.
Why did they need WHMFC?
In order to accurately determine the anisotropic parameters of the Cr(II) center, the high frequency electron paramagnetic resonance (HF-EPR) experiments were carried out at 4.2 K on the polycrystalline powder samples. The least-square fit to a complete two-dimensional array of the resonances gives the full set of spin Hamiltonian parameters, with S= 2,|D| = 2.15(3) cm-1,E= 0.01(1) cm-1,gx=gy= 2.02(2),gz= 1.90(5). Simulations to the 170 GHz spectrum indicate the sign of D is negative.
Who did the research?
Wenqing Wang,†,§Jing Li,†,§Lei Yin,‡Yue Zhao,†Zhongwen Ouyang,‡Xinping Wang,*,†Zhenxing Wang,*,‡You Song,*,†Philip P. Power*,ǁ
†State Key Laboratory of Coordination Chemistry, School of Chemistry and Chemical Engineering,Collaborative Innovation Center of Advanced Microstructures,Nanjing University, Nanjing 210093, China
‡Wuhan National High Magnetic Field Center & School of Physics, Huazhong University of Scienceand Technology, Wuhan 430074, China
ǁDepartment of Chemistry, University of California, Davis, CA 95616, USA
§ These authors contributed equally.
